Battling bad bugs
University of Guelph scientists fight antibiotic resistance by using our synchrotron to study scab disease in potatoes.
By Colleen MacPhersonScabin crystals as seen under a microscope.

Scientists fight antibiotic resistance by using synchrotron to study scab disease in potatoes.
In the ongoing war against antibiotic resistant bacteria, a change in battle tactics may prove effective for controlling a common disease of plants and potentially other toxins that affect humans and animals.
Although bacterial toxins cause serious, often deadly diseases, “bacteria aren’t trying to be nasty,” said Dr. Rod Merrill, Professor of Molecular and Cellular Biology at the University of Guelph. “They’re hungry and looking for food, and we’re often the food.” He added that 99 per cent of bacteria are helpful – like gut flora – so the battle is against the remaining one per cent.
The usual approach is to develop antibiotics “that kill the bacteria but not us, or the plant, or the animal,” stated Merrill. However, bacteria mutate quickly, as quickly as every 30 minutes, which leads to antibiotic resistance. “And unfortunately, the pipeline for new antibiotics is empty.”

The approach that Merrill and his research group are pursuing is an anti-virulence strategy – finding or designing small molecules that inhibit the tools bacteria use to colonize the host and create infection. “If we can put a lock on their weapons, they can’t get food and will move on so there’s not the same pressure to mutate. We’re going with this approach because we think it’s time to change up tactics.”
To validate the concept, Merrill turned to a bacterium called Streptomyces scabies that causes the common scab disease in root and tuber vegetables like potatoes and carrots. The disease creates lesions on the vegetables, making the food unsightly and unsuitable for sale.
“Potato is the most important vegetable crop in Canada and the fourth largest crop in the world,” said Merrill, who grew up on a beef farm in southern Alberta. Since there is no treatment, common scab has a significant economic impact for producers, reduces the arable land available for growing because it persists in soil, and creates potential food security issues for consumers.
Merrill and his team used the Canadian Light Source (CLS) at the University of Saskatchewan to study a protein toxin produced by the Streptomyces scabies pathogen. The CMCF beamline at the CLS helped them to generate a 3D structure of the toxin and to identify the active-site of the protein, which he described as “the mouth of the wolf.” The toxin uses its active-site to bind and cleave cellular molecules which damages the healthy host cells. It was critical information in finding a small-molecule inhibitor “to muzzle the monster.”
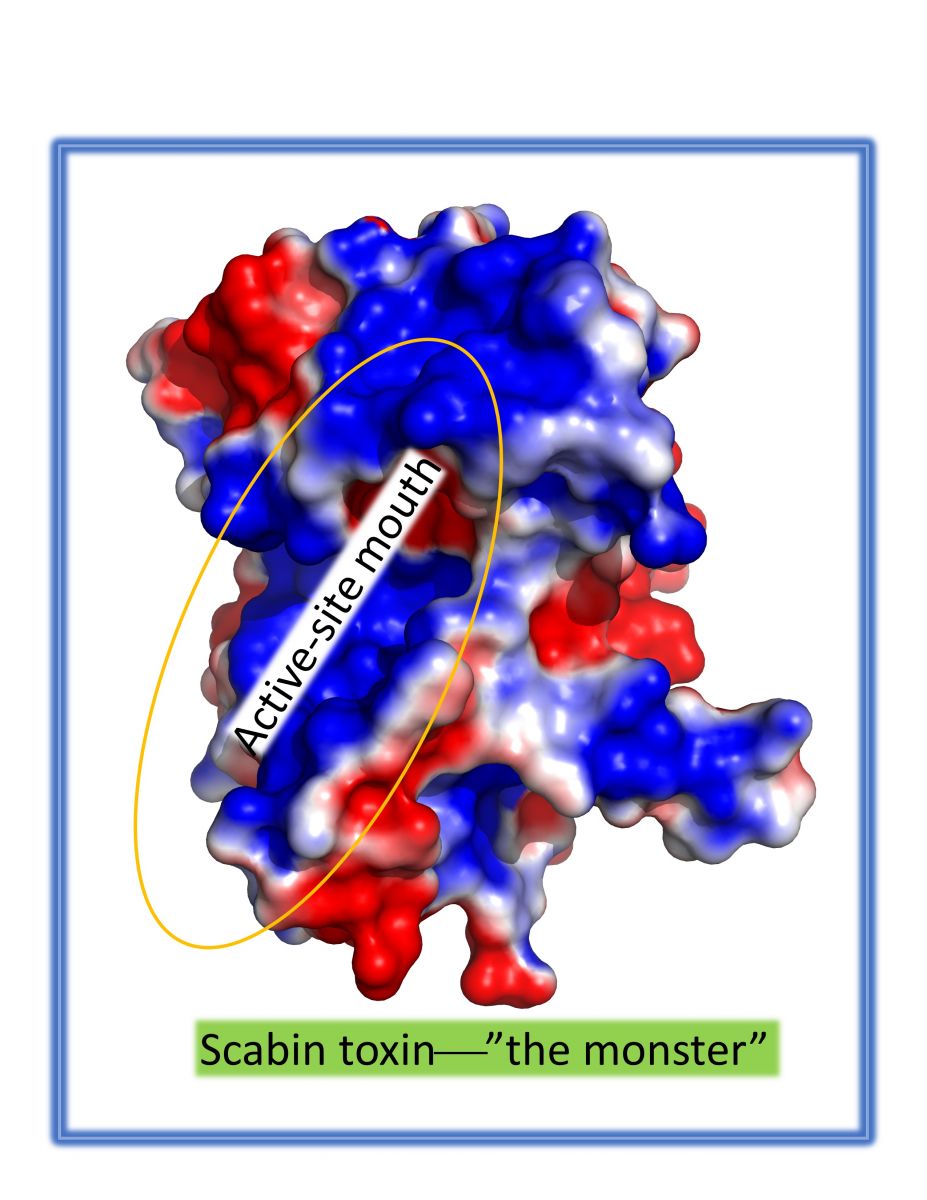
However, there is still further work required to demonstrate that the toxin inhibitors will work to neutralize the Streptomyces scabies pathogen. Once the anti-virulence approach has been established in the laboratory environment, the questions become, “is it practical, at what cost and will it work in a crop field environment? We haven’t yet demonstrated that it will work in cell cultures, but ultimately, we hope to work with agriculture specialists who can make a viable product to help farmers. I would love to see this go to the next level.”
Merrill believes the anti-virulence strategy in the fight against bacterial infections has potential applications far beyond diseases of root vegetables, including infections in humans, an area to which he devoted 15 years of his career.
Human medical research is rewarding, but finding trigger locks for the weapons of pathogens that affect animals, plants and his current specialty, honey bees, offered the opportunity to return to his agricultural roots.
“It’s very fulfilling working with the bee-keeping industry, and on other crop and animal diseases.”

Vatta, Maritza, Bronwyn Lyons, Kayla A. Heney, Taylor Lidster, and A. Merrill. "Mapping the DNA-Binding Motif of Scabin Toxin, a Guanine Modifying Enzyme from Streptomyces scabies." Toxins 13, no. 1 (2021): 55. DOI: 10.3390/toxins13010055.
To arrange an interview, contact:
Victoria Schramm
Communications Coordinator
Canadian Light Source
306-657-3516
victoria.schramm@lightsource.ca
