XAS
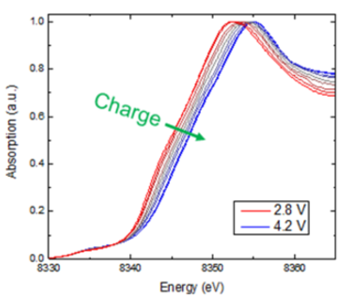
X-ray absorption spectroscopy (XAS) provides information on the chemical and electronic environment of specific elements, which can be used to identify different chemical species present in a sample. Some information is provided directly (like the oxidation state of a particular element) while other information can be obtained using fingerprinting methods and standards. Some examples of how we use XAS include determining the species of arsenic present in mine tailings and monitoring the oxidation/reduction of elements in a battery electrode.
Energy Range: 5.5-40,000 eV
Applications:
CROP DEVELOPMENT AGRI-PRODUCTS ENERGY MATERIALS CATALYSTS MINING BIOMEDICAL IMAGING
Loading...
XRF
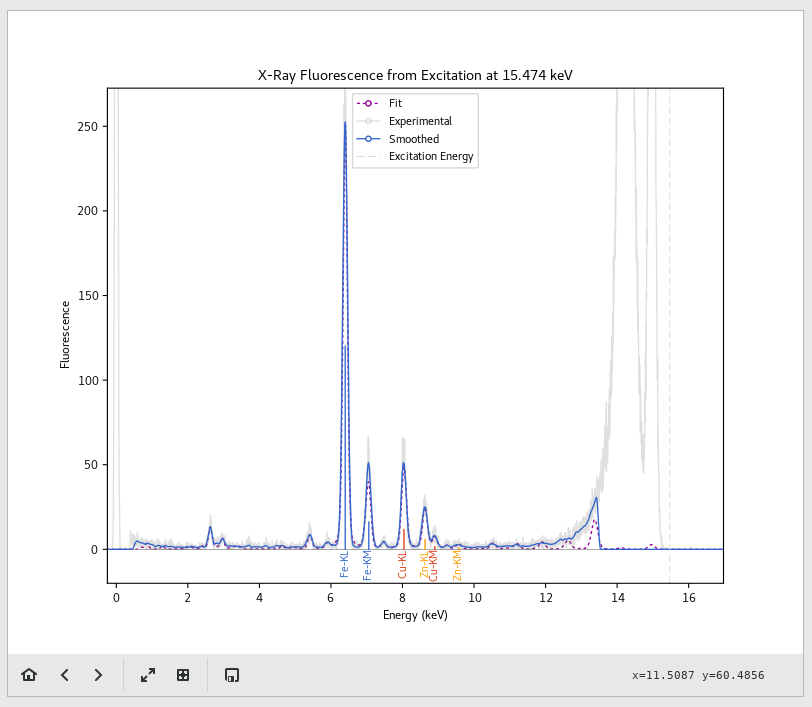
X-ray Fluorescence (XRF) is a common laboratory technique for determining the relative amounts of different elements present in a sample. By focusing a synchrotron beam down to a very small spot and measuring the resulting XRF pattern given off by the sample, an elemental map can be created for multiple elements simultaneously. These maps are highly sensitive and are extremely useful for correlating the distribution of elements in a sample.
Applications:
CROP DEVELOPMENT AGRI-PRODUCTS ENERGY MATERIALS CATALYSTS MINING BIOMEDICAL IMAGING
Loading...
XPS
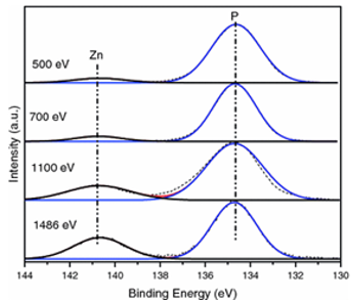
X-ray Photoelectron Spectroscopy (XPS) is a common laboratory technique for analyzing the surface chemistry of samples. XPS is element-specific and can be used to track changes in chemistry (like oxidation state) of elements that are very close to the surface (typically within a couple of nanometers). The spectral resolution of synchrotron XPS is significantly better than that of laboratory sources, which allows for separation of chemically similar phases. The wavelength of synchrotron X-rays can also be continuously tuned, which facilitates changing the penetration depth of the beam to profile different layers near the surface (up to a maximum of 70 nm depending on the material).
Energy Range: 240-10,000 eV
Applications:
ENERGY MATERIALS CATALYSTS
Loading...
XES
X-ray emission spectroscopy (XES) is a form of X-ray spectroscopy in which electrons are excited out of their shells and the emitted photons of the recombinating electrons are measured. While Resonant Inelastic Scattering (RIXS) is scattering in the sense that it is inelastic and appears relative to an elastic emission line, it is spectroscopic in nature as energy is lost through an electronic transition via an intermediate state, and both elastic and inelastic scattering can be measured with a spectrometer during XES experiments, along with fluorescence. XES is both element- and site-specific, making it a powerful tool for determining detailed electronic properties of materials.
Energy Range: 100-2,000 eV; 8-50 keV
Applications:
ENERGY MATERIALS CATALYSTS
Loading...
ARPES

Angle-Resolved Photoemission Spectroscopy (ARPES) is based on the photoelectric effect, in which an incoming photon of sufficient energy ejects an electron from the surface of a material. The kinetic energy and emission angle distributions of the emitted electrons are then measured to map the elecronic band structure and Fermi surfaces of the sample. The CLS has capabilities for both angle-resolved photoemission (ARPES) and spin-resolved photoemission (SARPES) experiments.
Energy Range: 15-10,000 eV
Applications:
CROP DEVELOPMENT AGRI-PRODUCTS ENERGY MATERIALS CATALYSTS MINING
Loading...
XEOL
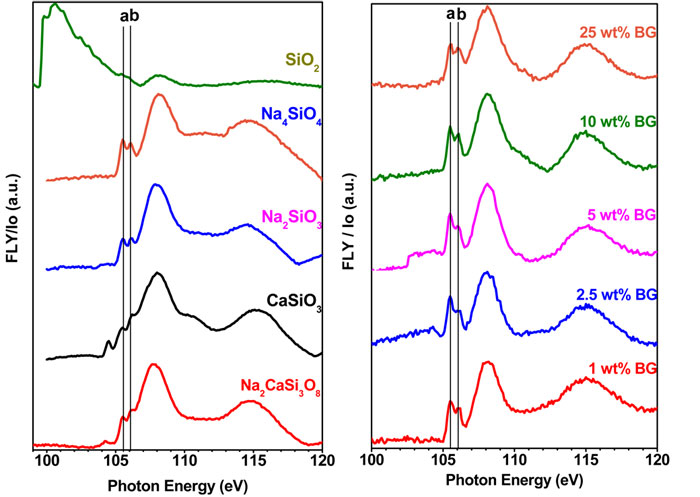
X-ray excited optical luminescence (XEOL) is a photon in - photon out technique. The absorption of photons produces energetic electrons which then cause further excitation and ionization, resulting in the release of photons. Photons in the visible range are detected as XEOL while emitted x-rays are measured as the Fluorescence Yield (FLY). Time-resolved XEOL and microprobe capabilities are also available. XEOL can be performed in a variety of sample environments including ambient and high-pressure conditions, or using an in-vacuum lens in a solid state chamber.
Energy Range: 55-30,000 eV
Applications:
CROP DEVELOPMENT AGRI-PRODUCTS ENERGY MATERIALS CATALYSTS MINING
Loading...
FTIR
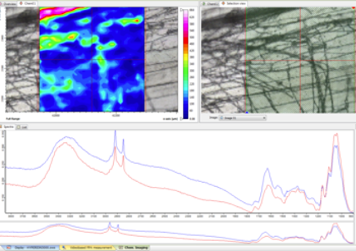
Fourier Transform Infrared Spectroscopy (FTIR) is a technique that involves passing infrared light through a sample. Some photons are absorbed by the sample and some are transmitted. A spectrometer collects high-resolution spectral data over a wide spectral range, and a mathematical process called a Fourier transform is used to convert the raw data into a spectrum. FTIR can be used to study and identify chemical substances or functional groups in solid, liquid, or gaseous forms.
Energy Range: 5-6000 cm-1
Applications:
CROP DEVELOPMENT AGRI-PRODUCTS ENERGY MATERIALS BIOMEDICAL IMAGING
Loading...
Access
Purchased Access
Purchased access offers quick and accurate solutions to proprietary questions. CLS scientists develop experimental plans based on clients' needs, collect and analyze data, and provide detailed reports with key answers to critical questions.
Peer-Reviewed Access
Academic clients can submit proposals through a peer review process. Beam time is granted based on scientific merit, with the expectation that any results will be published. In special cases, rapid access is also available for instrument or beam time.
